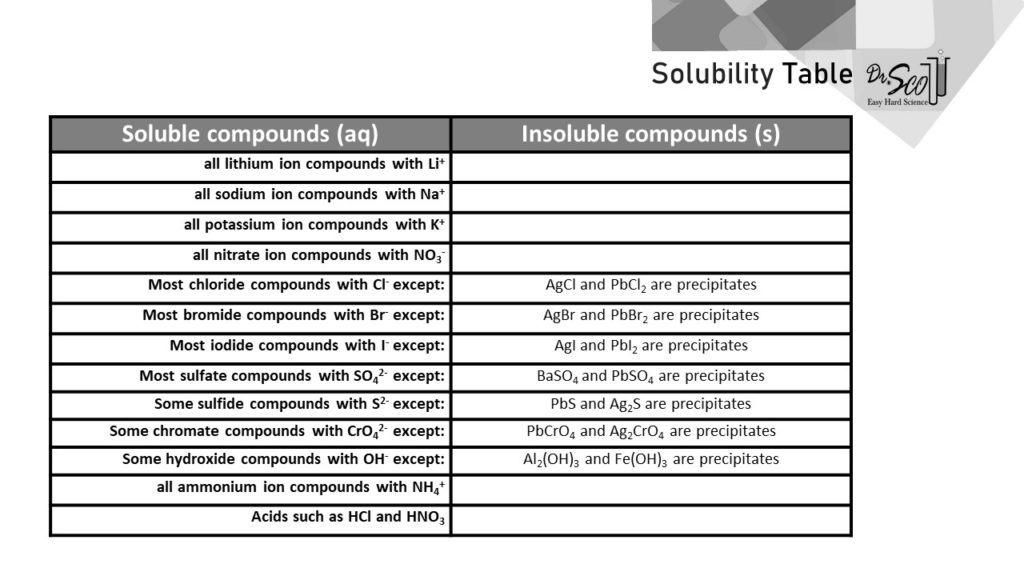
What Information Does a Solubility Table Give
And determines the yield. This is the solubility table that is available for use within ALEKS.
Solubility Table From A Chemistry Book Download Scientific Diagram
Its possibly easier to memorise the insoluble substances than the soluble ones.
. The solubility curve plots the changes of the solubility of a solid at different temperatures in a solvent. --Itub 1830 26 March 2007 UTC Mass is. T25 C273 K 29815K.
Nitroethane and 1-butanol have the same. In general I tell students to start by memorising the 5 insoluble Chlorides and Sulfates. From the solubility information in the table below prepare a solubility curve graph for BaCl_2.
The solubility of metal cations in water is strongly influenced by the hydration. On a graph the variations in temperature are plotted on the X-axis and the solubility is plotted on the Y-axis. For any drug its solubility is an important physical chemical property.
It tells which compounds will dissolve in water. 316 357 407 466 526 588 Outline a As the temperature increases does the solubility of BaCl2 in water increase or decrease. Solubility tables tables of solubility usually tabulate the maximum mass in grams of the solute that can be dissolved in 100 mL of water.
Solubility and Solids. What information does solubility table give. When mixed with other ions they can become precipitates or remain aqueous.
A solubility chart with a list of ions. Commonly solubility data is given for 25C. The following chart shows the solubility of multiple independent and various compounds in water at a pressure of 1 atm and at room temperature approx.
What information does a solubility table give. From the solubility information in the table below prepare a solubility curve graph for BaCl2. It tells which compounds will dissolve in water.
Even so you will save a lot of time now and later in UT Chem classes if you commit these two solubility rules to memory. The Hildebrand solubility parameter can be useful but it does not account for the origins of molecular stickiness or their consequences This means it is possible for various combinations of intermolecular interactions to give rise to the same Hildebrand solubility parameter EXAMPLE. The table below provides information on the variation of solubility of different substances mostly inorganic compounds in water with temperature at one atmosphere pressureUnits of solubility are given in grams per 100 millilitres of water g100 mL unless shown otherwise.
Interactive and user-friendly interface. The solubility table shows you a list of salts that are soluble and insoluble. The solubility of a substance in a particular solvent is measured by the concentration of the saturated solution.
Compounds with Group 1 cations and NH 4. When mixed with other ions they can become precipitates or remain aqueous. Thats why many solubility tables give the solubility per mass of solvent rather than volume.
Therefore knowledge of the solubility is essential for the design of the crystallization process. At 25C and 1013 kPa 1 atm the density of water is 100 g mL-1. The solubility of a substance fundamentally depends on the solvent used as well as temperature and pressure.
Propose an explanation for the observed solubility pattern. You may have noticed that you can dissolve more sugar or table salt in hot water than in cold water. Mass does not change with fluctuations in temperature The mass of a fixed volume of liquid does change with temperature.
The size or radius of an atom increases as one goes down a row family in the periodic table. It is one of the important parameters that will help you to achieve the desired concentration of drugs in systemic circulation for the most anticipated pharmacological response. It tells which reactants are in the gaseous state.
B What is the solubility of BaCl2 at 50C. It tells which compounds react with oxygen. As the temperature increases does the solubility of BaCl_2 in water increase or decrease.
Temperature 0C 20C 40C 60C 80C 100C g BaCl2 100 g H20. A solubility chart with a list of ions. What information does a solubility curve provide.
The values at STP will be. When mixed with other ions they can become precipitates or remain aqueous. Soluble - soluble more than 1g per 100g of water low - low solubility 001g to 1g per 100g of water insoluble - insoluble less than 001g per 100g of water.
Solubility is the ability of a solid liquid or gaseous chemical substance referred to as the solute to dissolve in solvent usually a liquid and form a solution. Ingredients AI the solubility curve helps to choose a suitable crystallization process cooling or evaporative crystallization eg. A solubility chart with a list of ions.
What information does a solubility table give. The substances are listed in alphabetical order. The following chart shows the solubility of multiple independent and various compounds in water at a pressure of 1 atm and at room temperature approx.
Feel free to have this table with you when you take the assessment or when you are in learning mode in ALEKS. Temperature and the solubility of salts. Once you can recite that list off the top of your head then add in most carbonates.
N denotes number of moles of gas. Solubility is the phenomenon of dissolution of solute in the solvent to give a homogeneous system. We use Flash technology.
The possibility of studying the gaming table. Using the graph find the solubility of BaCl_2 at 50 degree C. This data in the table is relevant only to the temperature given for the table.
The following chart shows the solubility of multiple independent and various compounds in water at a pressure of 1 atm and at room temperature approx. A new and reliable information on the solubility of salts acids and bases. Design experiments in the virtual lab to answer the follow questions.
For cations the charge density charge per unit volume will therefore decrease going down a row. It tells which metals will replace other metals. R denotes gas constant.
Solubility curve provides us information about how many gram of salt can be dissolved in water at different temperature. Measuring the solubility requires accurate control of temperature. This is because the solubility of these substances increases with temperature.
Temperature plays an important role in solubility because the solubility of a substance is different at different temperatures. It tells which compounds will dissolve in water. It tells which compounds will dissolve in water.

Solubility Rules Chart Chemistry Chemtalk
Comments
Post a Comment