Which of the Following Properties of Water Involve Hydrogen Bonds
E The very low molecular weight of water. C Hydrogen bonds can form between neighboring molecules.
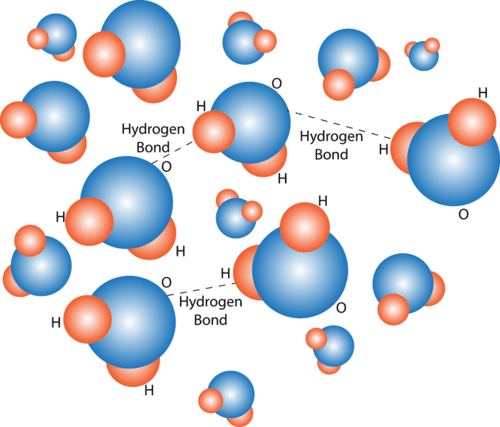
Biochemical Properties Of Water Advanced Read Biology Ck 12 Foundation
Water is one of the things that help in the existence of life on the earths surface.
. Water has a ____ surface tension allowing adhesion cohesion and capillary action. Higher density of liquid versus solid 1 only 2 only 3 only 1 2 and 3. Theattraction between individual water molecules creates a bond knownas a hydrogen bond.
Hydrogen bonds explain which of the following properties of water. Hydrogen bonds have an essential role in drug discovery as most oral drugs have about 8-10 hydrogen bonds. When heat is absorbed hydrogen bonds are broken and water molecules can move freely.
All of these properties. Water has a high heat capacity and heat of vaporization and icesolid wateris less dense than liquid water. Both the abnormal melting and freezing points and that ice expands when frozen.
A Hydrogen atoms covalently bond with each other to create one stable valence shell of electrons. Which of the following properties of water involve hydrogenbonds. Which of the following properties of water involve hydrogen bonds.
The hydrogen bond is longer than a normal covalent bond. Here are the Properties of water quiz questions and answers. Lower alcohols are soluble in water because of the hydrogen bonding which can take place between water and alcohol molecule.
When the temperature of water decreases the hydrogen bonds are formed and release a considerable amount of energy. See the answer See the answer done loading. D The density of water is greater than the density of ice.
Chemistry questions and answers. It is generally 20 times weaker than a covalent. Properties of hydrogen bond.
B High heat of vaporization. B One oxygen atom transfers an electron to each of two hydrogen atoms forming an ionic bond. Water has high melting and boiling points for its small size.
This quiz covers the characteristics of water and the importance of these characteristics to. E water expands as it freezes. As the compounds involving hydrogen bonding between different molecules have a higher boiling point so they are less volatile.
Water is a great solvent for all ionic and polar molecules. Ice expands when frozen. The hydrogen molecule then forms a hydrogen bond with an oxygen atom to create the water molecule.
Check all that apply. High melting point 2. At its boiling point of-100 C 207 kJ of heat is required to vaporize 532 g of sulfur dioxide.
The unique physical properties including a high heat of vaporization strong surface tension high specific heat and nearly universal solvent properties of water are also due to hydrogen bonding. Cohesion and adhesion of water. High surface tension 3.
E Hydrogen bonds are responsible for many of the properties of water. Waters high heat capacity is a property caused by hydrogen bonding among water molecules. The hydrophobic effect or the exclusion of compounds containing carbon and hydrogen nonpolar compounds is another unique property of water caused by the hydrogen bonds.
Nonpolar AND Polar Covalent. High heat of vaporization. Low vapor pressure 4 high surface tension a 1 and 3b2 and 3 c2 3 and 4 d 1 3 and 4 e 123 and 4 2.
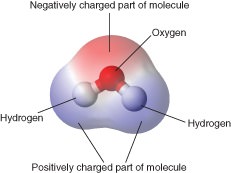
Water owes these unique properties to the polarity of its molecules and specifically to their ability to. C High specific heat. None of these properties.
Hydrogen Bonds typically occur between atoms of hydrogen involved in this type of bond. Check all that apply. The human body uses water in all its cells organs and tissues to help regulate its temperature and maintain other bodily functions.
E water expands as it freezes. Even though it requires energy to change a liquid to a gas some water molecules break their hydrogen bonds at ordinary temperatures by. High heat of vaporization 3.
In water each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. In the case of water hydrogen bonds form between neighboringhydrogen and oxygen atoms of adjacent water molecules. Which of the properties of water result from hydrogen bond formation.
Hydrogen bonding in water is responsible for different properties of water like high boiling point surface tension and solvent nature. B high heat capacity. High heat of vaporization Surface tension Adhesion Cohesion High specific heat.
The hydrogen bond is stronger than dipole-dipole interaction but weaker than the normal covalent bond. Hydrogen bonds explain all of these properties. Water is a polar molecule.
A Cohesion of liquid water due to hydrogen bonding. Which of the following properties of water does not contribute to the fitness of the aqueous environment for living organisms. Which of the following properties of water is related to its ability to form Hydrogen or H-bonds.
E water expands as it freezes. Water readily dissociates ionic compounds dissolves covalent molecules and hydrates the smaller particles. B high heat capacity.
Properties of Hydrogen Bonding. In H 2 O only two of the six outer-shell electrons of oxygen are used for this purpose leaving four electrons which are organized into two non-bonding pairs. Water can stick to itself cohesion and other molecules adhesion.
Check all that apply. B high heat capacity. D Hydrogen bonds are important for holding large molecules together.
Which of the following properties of water can be attributed to hydrogen bonding. High heat of vaporization 2. Check all that apply.
Which of the following properties of water can be attributed to hydrogen bonding. This is most likely because. Up to 256 cash back These attractions are much weaker than true ionic orcovalent bonds but they are strong enough to result in someinteresting properties.
1 Water the molecule. Which of the following properties of water involve hydrogen bonds. Waters boiling point is ____ abnormally than expected.
A Hydrogen bonds are strong attractive forces between hydrogen atoms and negatively charged atoms. Specific heat heat of vaporization and density of water.
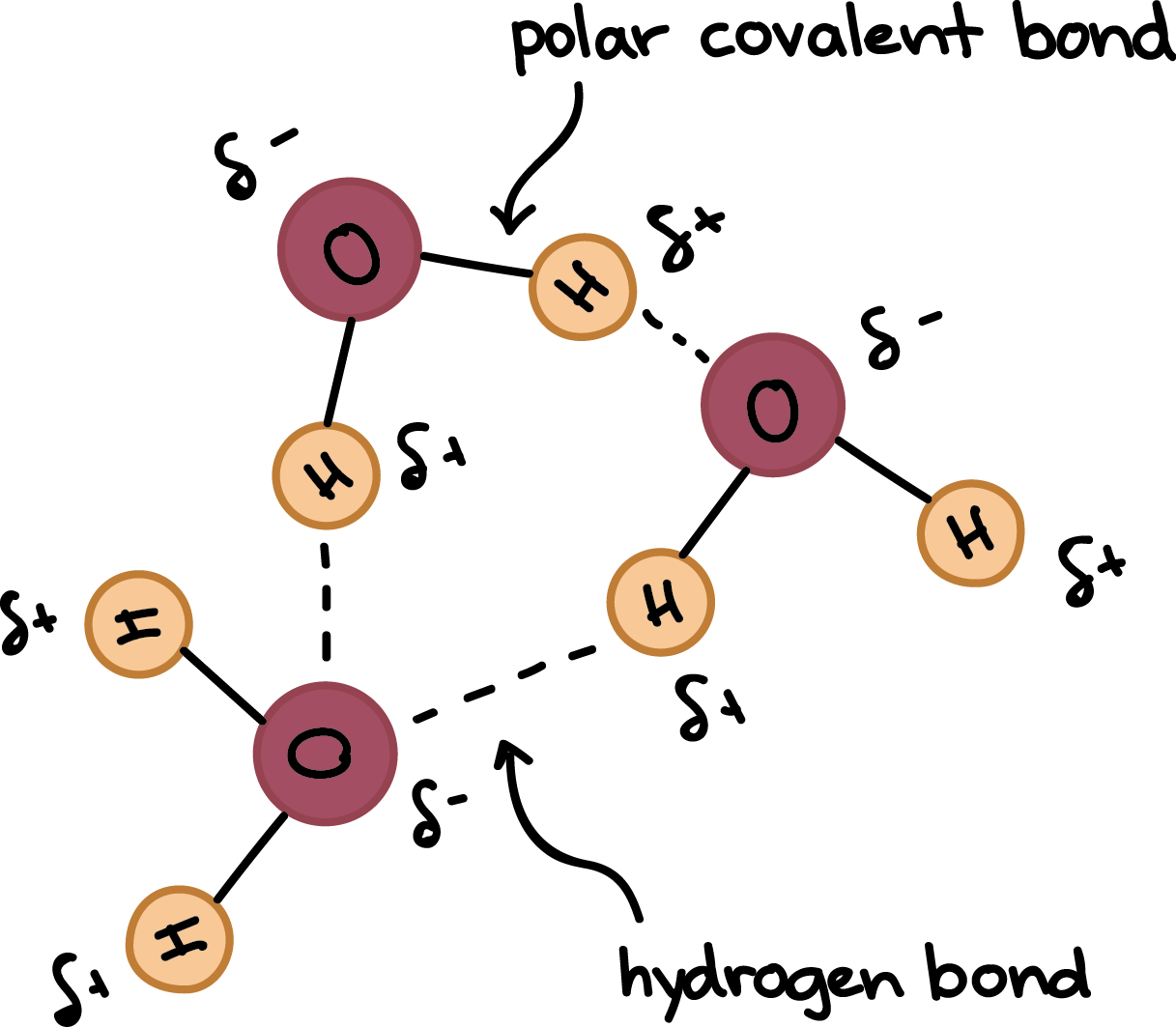
Hydrogen Bonds In Water Article Khan Academy
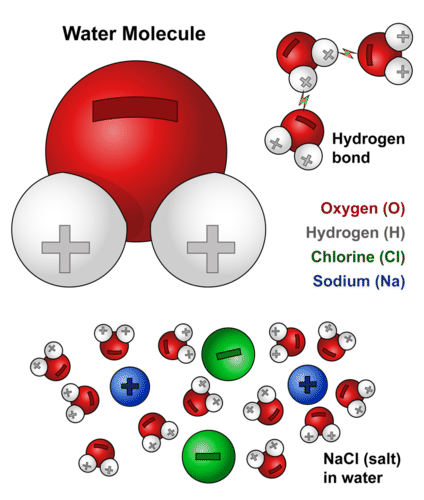
Biochemical Properties Of Water Advanced Read Biology Ck 12 Foundation

Biochemical Properties Of Water Advanced Read Biology Ck 12 Foundation

Comments
Post a Comment